This page was produced as an assignment for Genetics 677, an undergraduate course at UW-Madison.
Protein Structure
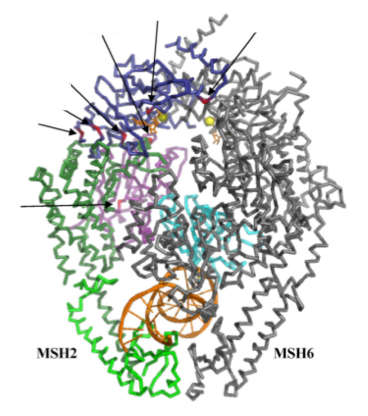
The x-ray crystal structure of MSH2/MSH6 dimer holding a DNA helix is shown in Figure 1. A diagram showing the protein domain functions of the MSH2 protein is shown in Figure 2. MSH2 protein domains are color-coded so they can be visualized in the x-ray crystal structure.
Analysis
Protein domains are color-coded to visualize them in the x-ray crystal structure of the protein. All domains are seemingly interacting with either MSH6, DNA or ATP, indicating that most, if not all, regions of the MSH2 protein are vital for proper function of the MSH2 protein. Mutations in any of these domains may alter MSH2 binding, interaction, and function.
References
1. Anne Lützen, Niels de Wind, Dubravka Georgijevic, Finn Cilius Nielsen, Lene Juel Rasmussen. Functional analysis of HNPCC-related missense mutations in MSH2. Mutation Research. 2008. 645;44–55.